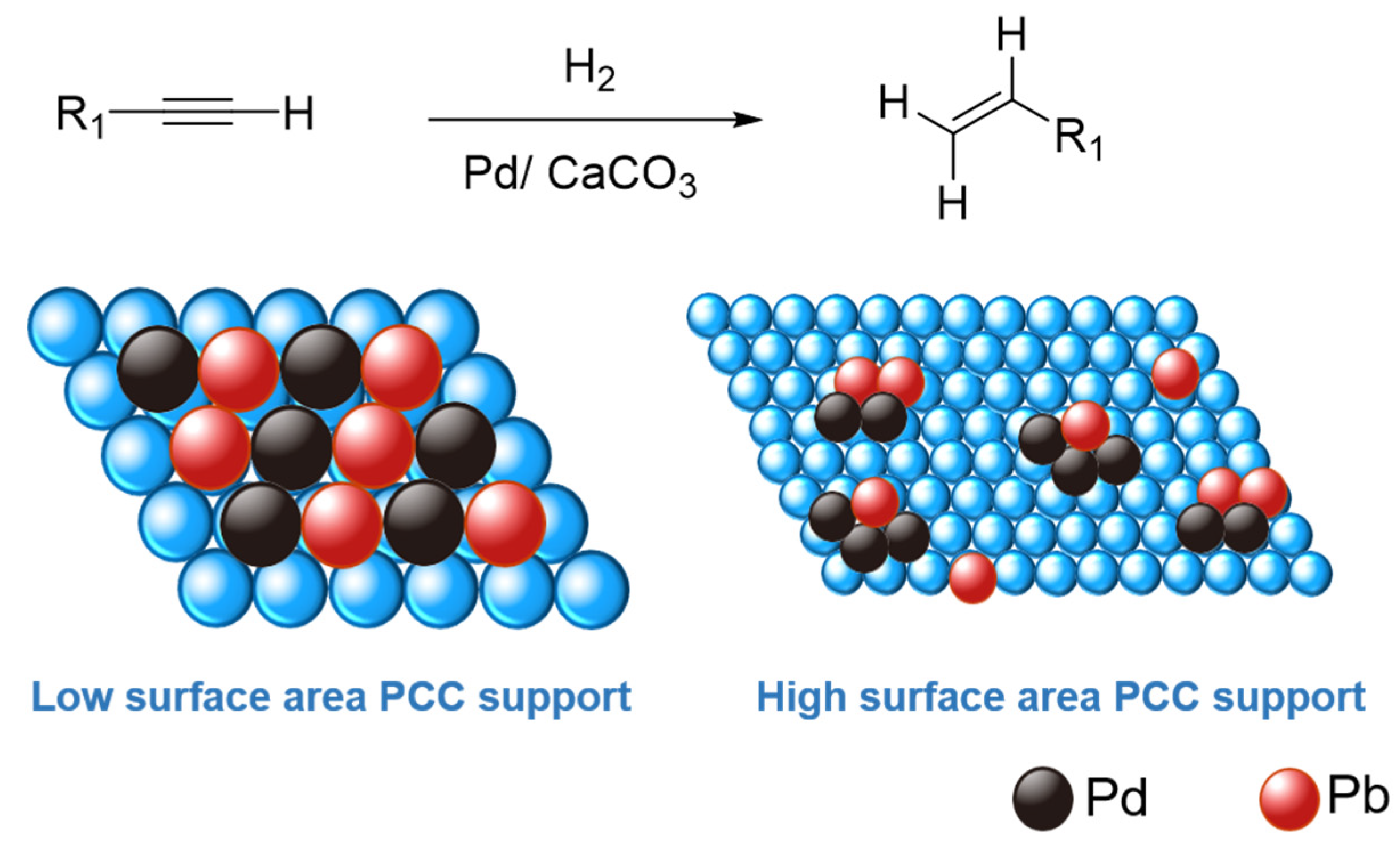
Palladium-catalyzed oxyarylation of olefins using silver carbonate as the base. Probing the mechanism by electrospray ionization mass spectrometry - ScienceDirect

Palladium-Catalyzed Deracemization of Allylic Carbonates in Water with Formation of Allylic Alcohols: Hydrogen Carbonate Ion as Nucleophile in the Palladium-Catalyzed Allylic Substitution and Kinetic Resolution | Journal of the American Chemical Society
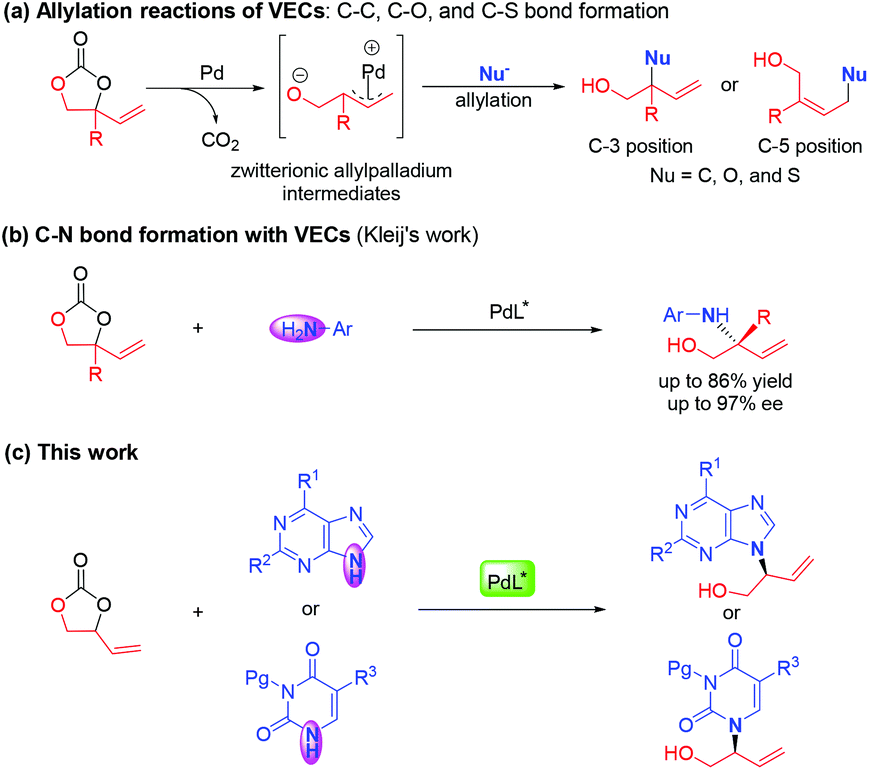
Palladium-catalyzed asymmetric allylic amination of a vinylethylene carbonate with N-heteroaromatics - Organic Chemistry Frontiers (RSC Publishing) DOI:10.1039/D1QO00272D

Reactivity of palladium(II) methyl complexes towards CO2: formation of carbonate complexes - ScienceDirect

Palladium-Catalyzed Decarboxylative Asymmetric Allylic Alkylation of Enol Carbonates | Journal of the American Chemical Society